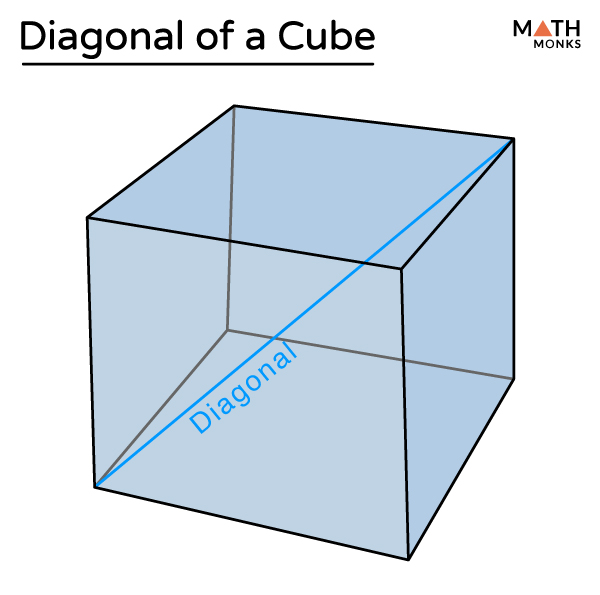
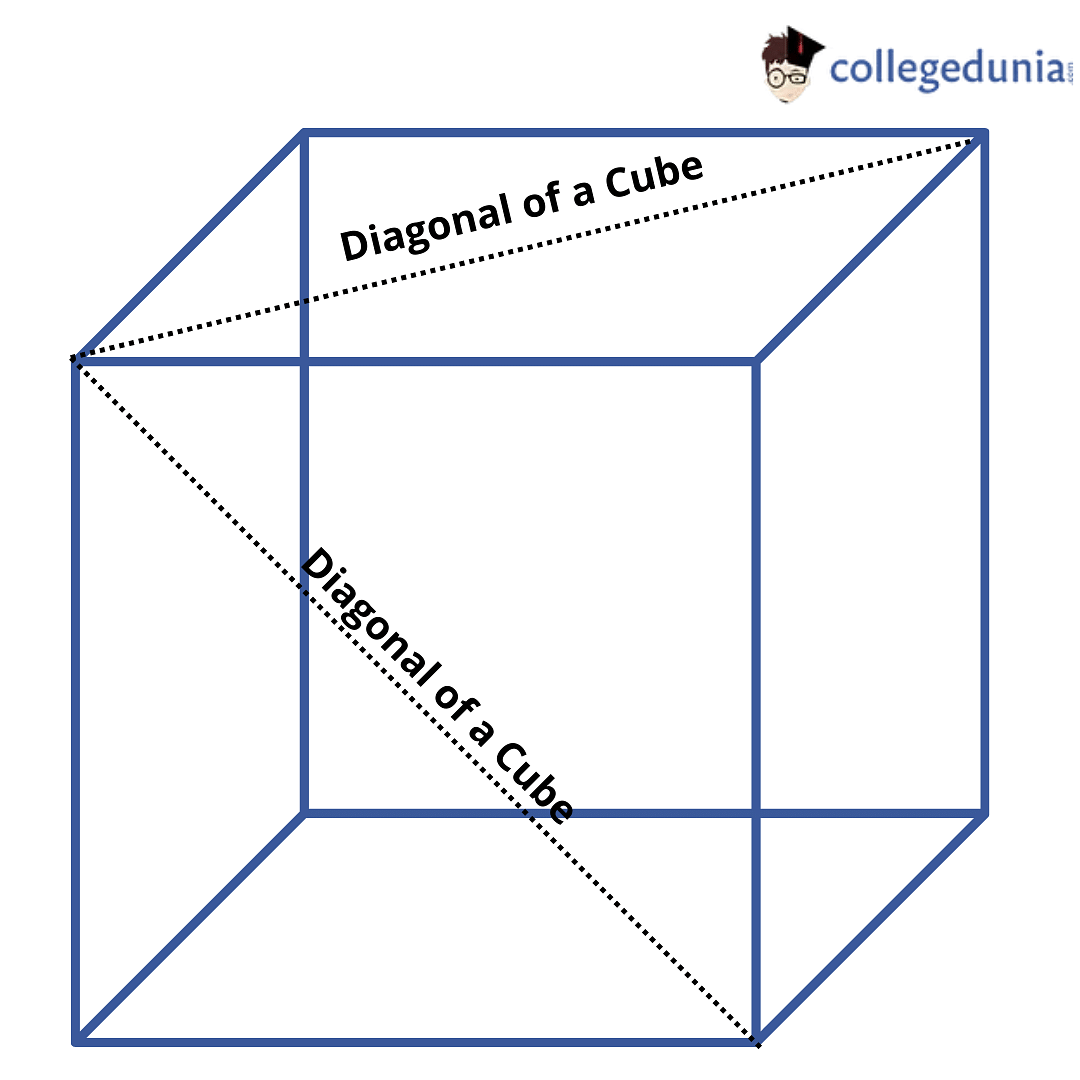
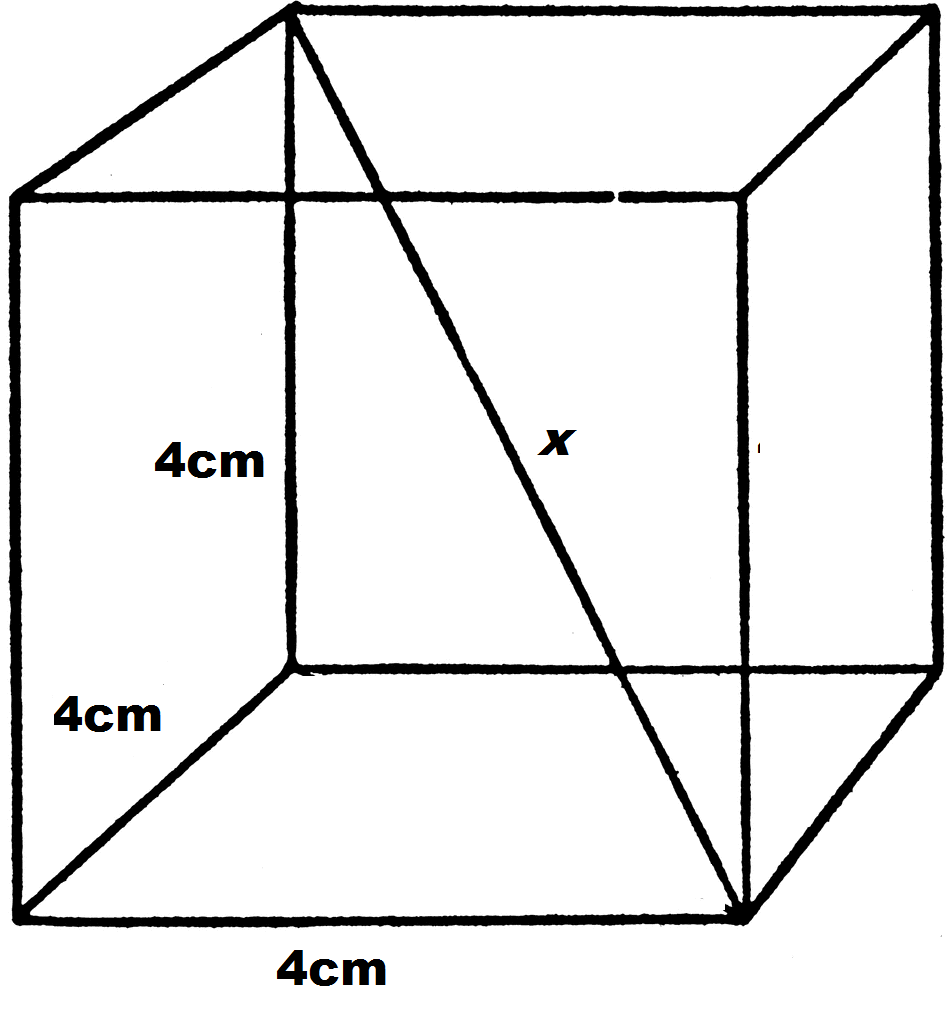
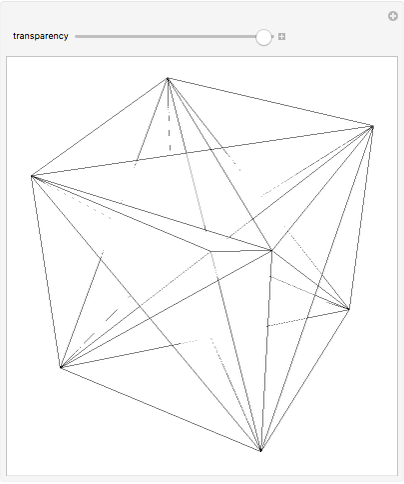
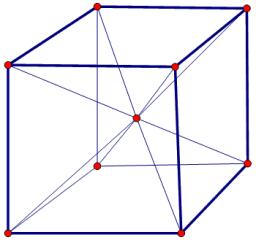
netry: One plane (b) Diagonal planes of symmetry: One passing diagonally through the cube is shown in Fig. 6.22 can be a total of 6 such planes passing diagonally through the che
Why do we consider the diagonal of a cube in the calculation of the edge length of a BCC crystal? - Quora
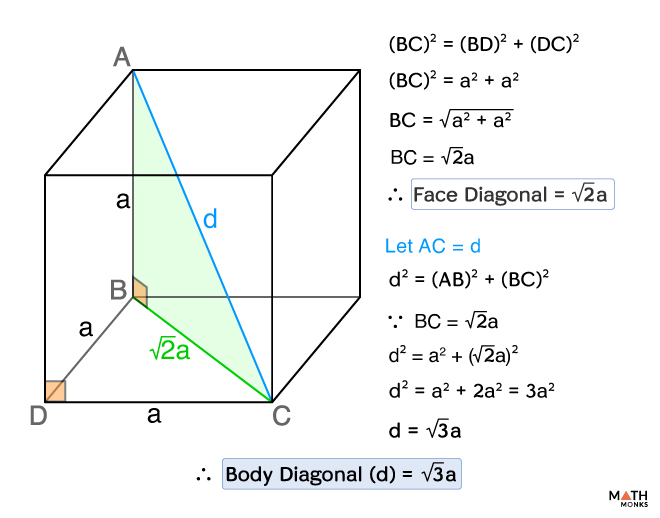
![If 'a' the length of side of the cube, the distance between the body-centred atom and one corner atom in the cube will be:[A] $\\dfrac{4}{\\sqrt{3}}a$ [B] $\\dfrac{\\sqrt{3}}{4}a$ [C] $\\dfrac{\\sqrt{3}}{2}a$[D] $\\dfrac{2}{\\sqrt{3}}a$ If 'a' the length of side of the cube, the distance between the body-centred atom and one corner atom in the cube will be:[A] $\\dfrac{4}{\\sqrt{3}}a$ [B] $\\dfrac{\\sqrt{3}}{4}a$ [C] $\\dfrac{\\sqrt{3}}{2}a$[D] $\\dfrac{2}{\\sqrt{3}}a$](https://www.vedantu.com/question-sets/720721fd-3ab5-4f3a-8864-68a0dfdd06901210945075913440462.png)
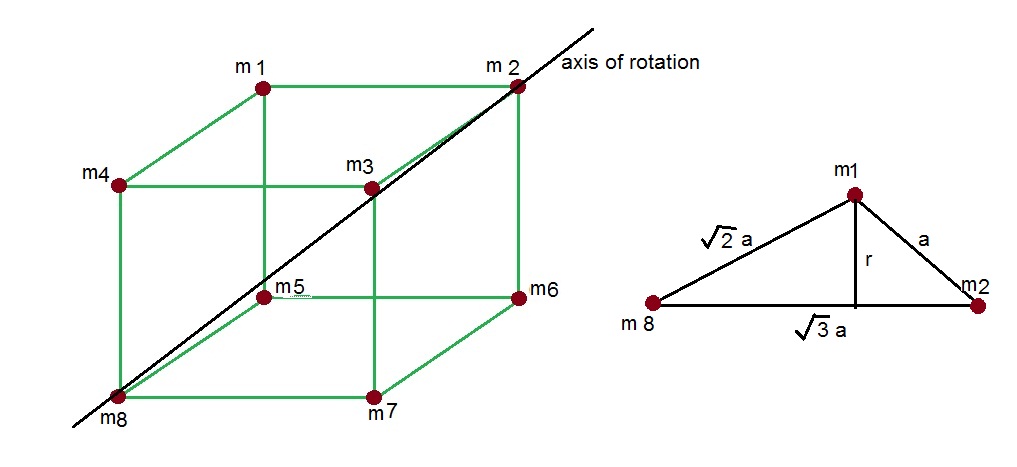
If 'a' the length of side of the cube, the distance between the body-centred atom and one corner atom in the cube will be:[A] $\\dfrac{4}{\\sqrt{3}}a$ [B] $\\dfrac{\\sqrt{3}}{4}a$ [C] $\\dfrac{\\sqrt{3}}{2}a$[D] $\\dfrac{2}{\\sqrt{3}}a$

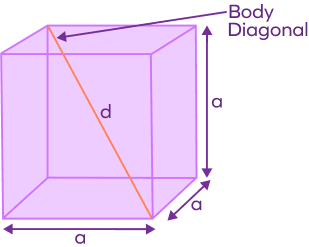
![Edge length of a cube is 400pm. Its body diagonal (in pm) would be:[A] 566[B] 600[C] 500[D] 693 Edge length of a cube is 400pm. Its body diagonal (in pm) would be:[A] 566[B] 600[C] 500[D] 693](https://www.vedantu.com/question-sets/4fe4a944-9f2c-437b-97ab-e350e21444618606447170386427521.png)